Segregation of bulk solids – causes and solutions
In many processes, individual steps involve the presence of bulk solids as the primary, intermediate or end product, and the individual particles can cover a size range encompassing eight powers of ten. The flow behaviour of bulk solids, however, depends not only on the size of the particles, but also on their shape and density and, not least, on the moisture content of the material. In certain applications, it is not only important to ensure reliable flow, i.e., to avoid flow obstructions, but also and primarily to prevent segregation.
1 What causes segregation?
Segregation becomes a problem when downstream processes require that the bulk solid be of uniform composition. This is the case, for example, when the product is to be packaged in portions of identical composition (sales packages or the like), or when a downstream process could become unstable if the product composition were to vary.
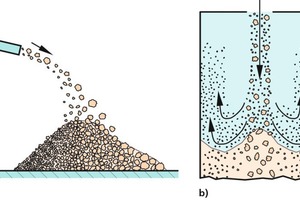
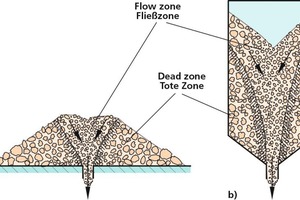
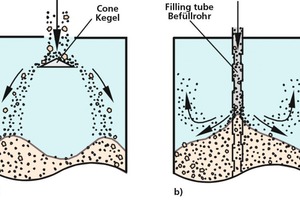
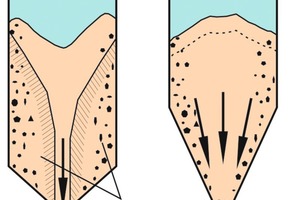
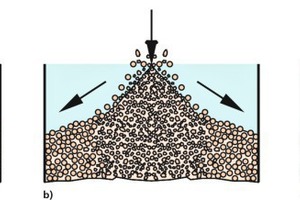
Bulk solids tend to segregate, if the particles differ in terms of size, shape or density. Bulk solids with very poor flow behaviour, such as very fine-grained titanium dioxide, or which are too moist (e.g., moist clay) normally show...

![»2 Steady-state settling velocity of spherical particles (calculated according to [11] for Re < 0.25 “Stokes law“)](https://www.zi-online.info/imgs/tok_2d96dff24531450b7878c7f052e54b62/w300_h200_x400_y295_101490946_d045448ebd.jpg)