“Natural Gas and Hydrogen – What’s New?” Findings from Laboratory and Field Studies (part 1 of 3)
1. Introduction and objective
The Brick and Tile Industry Roadmap 2050 points to ways to reduce energy consumption and CO2 emissions within the sector [1]. In recent years, particular attention has been given to green hydrogen as a substitute for natural gas. The use of this firing gas has raised questions among brick and tile makers with regard to its influence on the firing process and the end-product. With initial theoretical considerations and investigations on laboratory scale based on systematically adjusted kiln atmospheres, it has already been shown that hydrogen has no significant influence on the end-product [2], [3], [4].
It was not initially known how hydrogen used as a firing gas affects the industrial firing process and heavy clay products. For this reason, at the Weimar Institute of Applied Construction Research (IAB) in 2023, a research project was initiated in collaboration with an industry partner. In the scope of this project, raw materials and porosified clay bricks (HMz) were fired in different laboratory furnaces as well as in a tunnel kiln and the results compared with each other as well as with findings from previous investigations. In addition to comparing the product properties, the project was aimed at validating known theoretical insights like energy consumption, air and flue gas flows as well as emissions during firing with natural gas and hydrogen with the help of findings from field studies [5], [6].
To ensure the laboratory firings were as close as possible to industry firings, a chamber kiln was built at the IAB that can be flexibly heated with natural gas, hydrogen as well as freely definable mixes of these two firing gases. In this kiln, the industrial firing process could be simulated and, based on measurement of the gas volume and the flue gas composition, compared with the material outcomes. In addition, one objective was to clarify how far a raw-material-dependent percentage of harmful gas in the reduced flue gas volumes changes depending on the hydrogen content in the natural gas.
2. Kilns used
2.1. Electrically heated gradient kiln
To determine the suitable firing temperature of a material in oxidizing firing conditions, an electrically heated gradient kiln built by Linn Elektrotherm was used. The heating rate of this kiln was 200 K/h, cooling was unregulated. In one firing, 20 test specimens were fired at the same time at different temperatures and their ceramic properties subsequently determined.
2.2. Electrically heated hydrothermal kiln
For determination of the ceramic properties in different kiln atmospheres, firings were conducted in an electrically heated kiln supplied by MUT advanced heating (cf. [6]).
The kiln with heating coils on the inside has a maximum operating temperature of 1 200 °C. The interior chamber is gas-tight for firing in vacuum as well as for overpressure up to 2 bar. The double exterior wall is temperature-controlled to 130 °C with oil to avoid the formation of condensate. Air, nitrogen and carbon dioxide can be supplied to the kiln. At the kiln door, a nozzle can be used to supply water and nebulize this with the gas mix. The gas flows are adjusted with the help of a mass flow controller, while the water flow is varied with a liquid flow controller in the software for each segment of the firing curve. The total quantity of the wet gas mix is calculated from the sum of the individual gases and reached 1 200 standard litres per hour for all firings. The settings specified above enabled the reproduction of combustion atmospheres as found in gas-heated kilns.
2.3. Gas-heated chamber kiln
The field firings were performed in a gas-heated chamber kiln built by Bader und Scheffer Ofenbau, in which a brick stack measuring 500 x 500 x 500 mm³ can be fired. The kiln reaches a maximum temperature of 1 250 °C and can be heated with natural gas, hydrogen as well as any mix of the two gases. Besides featuring thermal afterburning and regulated cooling, the kiln is equipped with extensive measurement and control technology. Process control is enabled with the definition of 30 process steps. The measured temperatures, gas and air volume flows as well as the flue gas composition are recorded at one-second intervals.
The flue gas composition is determined with the SWG100 gas emission analyser manufactured by MRU. The flue gas is extracted through a heated hose direct at the kiln and is then dried. Electrochemical and NDIR sensors continuously determine the composition of the dry flue gas, and the gases carbon monoxide and dioxide (CO and CO2), nitrogen monoxide and dioxide (NO and NO2), methane (CH4), oxygen (O2) and sulphur dioxide (SO2) are detected.
2.4. Gas-heated tunnel kiln
The natural-gas-fuelled tunnel kiln in the brick plant was built in 1992 by the company Hans Lingl Anlagenbau. The kiln measures 151.5 m in length and is equipped with a fully automatic digital control system. A process control system regulates central parameters like push rate, gas supply, temperature and circulation rate. The temperature is monitored continuously by means of sensors along the length of the kiln.
Mainly surface ground HMz with a size of 373 × 175 × 249 mm are produced. The firing temperature of 890 °C and the firing time of 24 hours depend on the specific clay brick size and the setting mass. There are 53 kiln cars in the kiln with a lock at the kiln entrance and exit. Every car measures 5 100 × 2 800 mm² and is moved in two partial pushes. The distance between the firing holes is 1 400 mm.
The production rate is around 200 000 tonnes fired HMz clay bricks per year. The plant consumes around 4 mill. Nm³ gas per annum. This corresponds to CO2 emissions of around 8 000 tonnes per year.
3. Raw materials and conducted tests
For the material tests and chamber kiln firings, the raw materials from the brickworks were available. These were characterized based on their material composition (mineral content, grain size, inorganic oxides, organic components). The instruments used for this are described partly in [6]. The chemical analysis was performed with the Rigaku ZSX Primus IV as a sequential wavelength-dispersive X-ray spectrometer for quantitative element analysis in solids and powders. The total carbon and total sulphur analysis (C/S analysis) was conducted with the Eltra CS-2000 instrument. The analysis is performed depending on the specimen type in the resistance or induction furnace with downstream IR gas detection (IR absorption).
For the firings in the laboratory furnaces, two clays (T1 and T2) and two mixes (M1 and M2a) were used. The mixes were taken in 2023 from the production of low- (M1) and high-porosified products (M2a). From these materials, specimens were prepared and dried, For the firings in the gas and tunnel kiln, in 2024, original HMz clay bricks made from the high-porosified mix and dried in the brick plant were used (M2b). The firing conditions and the abbreviations used are shown in »Table 1.
The test specimens and clay bricks had the following dimensions:
Prisms L x W x H: 70 x 25 x 10 mm³
Plates L x W x H: 100 x 100 x 25 mm³
HMz L x W x H: 243 x 238 x 248 mm³ (ground)
In the gradient kiln, prisms prepared from all the raw materials were fired between 800 and 1 050 °C to enable comparison of their firing behaviour.
In the hydrothermal kiln and in the gas-heated chamber kiln, the following materials and test specimens were fired:
Prisms made of clay and the clay mixes
Plates made of the clay mixes
In addition, in the chamber and tunnel kiln, the original HMz from the brick plant were fired and then the compressive strength and bulk density were determined.
The firings in gas-heated chamber kiln were performed after another with natural gas and hydrogen on four unfired perforated clay bricks in each case. To determine the influence of the raw material reactions on the gas and air consumption as well as the flue gas rate and the emissions at the kiln, after that reference firings with natural gas and hydrogen were performed on already fired material. Reference fired ware was in each case one complete clay brick and three ground clay bricks after determination of the compressive strength. As a result of the determination of the compressive strength, a material loss was registered.
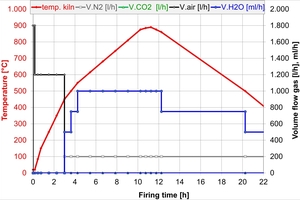
The firing curve in the hydrothermal kiln was generated on the basis of the investigations in [6] with the same kiln atmosphere as formed during the combustion of natural gas and hydrogen in the tunnel kiln. As the kiln atmospheres for hydrogen and natural gas in the tunnel kiln differ only slightly, two further firings were conducted – one with a high water vapour percentage and one in dry air. The water vapour atmosphere has a significant reducing effect, while the air atmosphere is oxidizing and corresponds to the electric heating of a kiln. The firing curve was the same for all firings in the hydrothermal kiln and is shown by way of example for the water vapour atmosphere in »1.
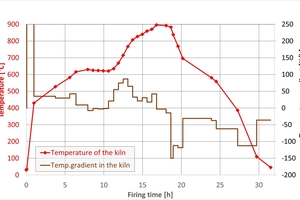
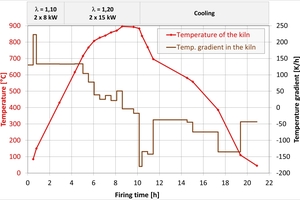
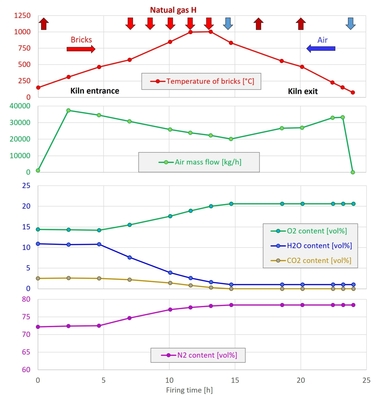
The actual firing curve in the tunnel kiln is shown in »2 left. Up to 600 °C a delay of six hours occurred as a result of a fault at the plant. The dwell time above 700 °C was around 6.6 h. The firing curve for the chamber kiln was the same for all firings and is shown in »2 right. It corresponds to the firing curve in the tunnel kiln that was shortened for laboratory conditions above 700 °C by the factor of 1.2. Up to the switchover point at 430 °C, the combustion air ratio lambda was = 1.10 and the output for each burner 8 kW. Above 430 °C, lambda was = 1.20 and the output 15 kW in each case. Cooling was effected with ambient air at the burner.
For the fired materials, the following characteristics were determined:
Prisms ceramic properties
Plates thermal conductivity
HMz compressive strength, bulk density, mineral content, chemical composition, carbon and sulphur content, pore structure, magnetic susceptibility
The ceramic properties of the prisms comprise the bending strength, shrinkage, mass loss, water absorption and the density. The shrinkage was measured with the help of shrinkage marks. Following determination of the bending strength in the three-point bending test, the water absorption and bulk density were determined by means of one-hour boiling of a test specimen half. The results are mean values from five individual test specimens in each case.
The compressive strength of the fired HMz was determined in accordance with DIN EN 772-1 in the brickworks. The values are means calculated from three individual values in each case.
The thermal conductivity was determined with the parallel plate method in accordance with ISO 8302, DIN 52612 and DIN EN 1946, Part 2 by means of TLP 900/100 – SG and the software Lambda V.2012, Two-plate from the company Taurus Instruments. For the measurement, the fired plates were ground to a uniform size. The thermal conductivity was determined from three individual measurements on two plates in each case.
The pore structure was determined on the tiles used in the thermal conductivity tests. With the scanning electron microscope Phenom ProX from the company Phenom World, images of the microstructure were captured with 500-, 1 000-, 2 000- and 5 000 x magnification. For the XRD measurement in reflection mode, the powder diffractometer STADI MP from STOE & Cie was used.
The mass-specific magnetic susceptibility was determined with the MS2 Susceptibility Meter from the company Bartington. The threefold determination was performed on 10 to 12 g specimen material in each case, prepared to be < 63 µm, which was filled and compacted into 12.5-ml specimen tins.
4. Findings
4.1. Composition of the raw materials
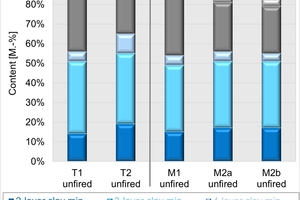
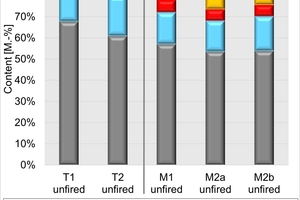
In the plant, two clays are used. The mixes consist of around 70 vol% of the two clays. They also contain different percentages of papermaking waste, bottom ash as well as mineral wool. The mineralogical and chemical composition of the unfired clays and mixes is shown in »3.
The mineralogical composition in »3 left shows that the Clay T1 is a high-quartz clay that contains kaolinite as well as illite and smectite. The clay mineral content of the schistous clay T2 is higher, in addition pyrite and organic components are present. Mix M1 has a higher content of quartz and somewhat lower contents of clay minerals and organics than Mix M2. Mixes M2a from 2023 and M2b from 2024 differ slightly in their composition despite having the same formulation. Mix M2b contains somewhat more organics and less quartz and calcite than Mix M2a. The clay mineral content of the mixes is almost identical.
The chemical composition in »3 right corresponds to the mineral content in respect of the contents of SiO2/quartz and CaO/calcite. The results of the C/S analysis confirm the contents of calcite, organics as well as pyrite in Clay T2 and the mixes.
4.2. Properties of the fired specimens
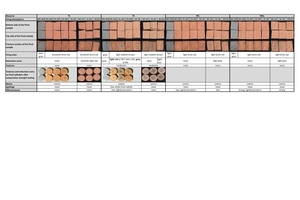
The fired colours and particular characteristics of the test specimens after firing are shown and described in »Table 2 (firing conditions and abbreviations cf. »Table 1).
The fired colours in »Table 2 show that in almost all firings the test specimens were coloured evenly brick red on the outside. A grey colouring was observed on all test specimens from the firing in water vapour atmosphere. Water vapour displaces oxygen in the kiln chamber almost completely, penetrates into the pores, reduces the iron minerals and leads to a grey colour. Slight differences in colour can be observed from material to material, which are attributed to the mineral content.
Reduction cores formed in Clay T2 in all kiln atmospheres, especially in the simulated natural gas firing in the hydrothermal kiln. In comparison with Clay T1, Clay T2 contains pyrite as well as organic components. The local oxygen consumption as a result of pyrite decomposition and the oxidation of the organics can lead to reducing conditions in the material, while the surface oxidizes in the kiln on account of the oxygen. The mixes on the other hand displayed light-coloured reduction cores, which were more pronounced in the chamber kiln than in the hydrothermal kiln. These are attributable to calcites and organics.
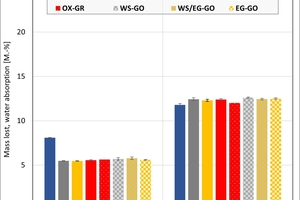
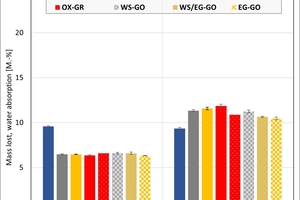
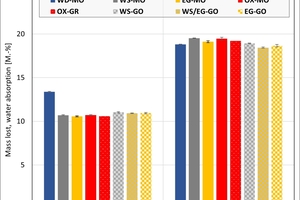
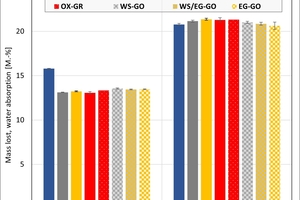
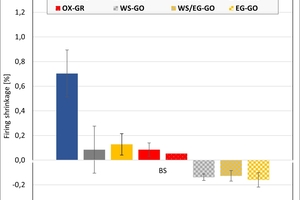
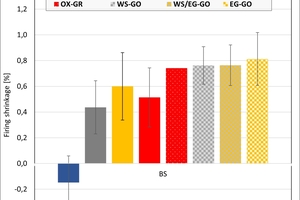
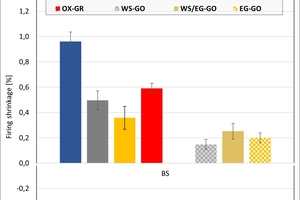
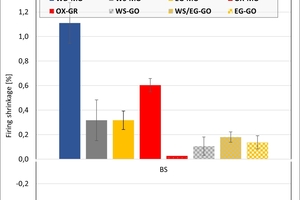
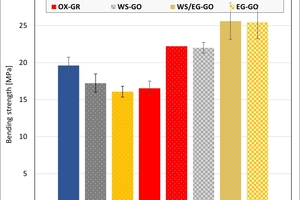
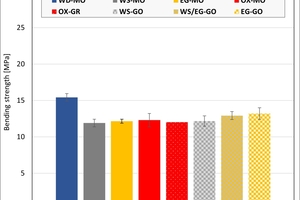
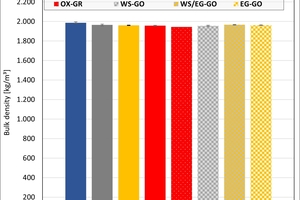
The ceramic properties after the firing in the different kilns and kiln atmospheres are shown in »4 to »7. In addition, the standard deviations from the mean of five measured values in each case are shown. The values from the gradient kiln are individual values without standard deviation.
The mass loss is almost the same for all kiln atmospheres – with the exception of the water vapour atmosphere, in which so far large unexplained losses occurred. In the chamber kiln, the mass losses are slightly higher than in the hydrothermal kiln. Clay T2 exhibits a larger mass loss than Clay T1 on account of its higher clay mineral content. The two mixes porosified (M1) and high-porosified (M2a) exhibit a larger mass loss than the individual clays on account of their content of calcite and organics.
The water absorption values show that the kiln atmosphere does not have any significant influence on the fired results. One exception is the water vapour atmosphere and especially Clay T2, which showed a much lower value than in the other atmospheres. In the chamber kiln, for both mixes somewhat lower water absorption values were achieved than in the hydrothermal kiln. The high water absorption values of the two mixes are caused by the calcite deacidification and the burnout of the organics.
The firing shrinkage of Clay T1 and the mixes is low and virtually independent of the kiln atmosphere. One exception is the water vapour atmosphere with predominantly higher shrinkage values. In the chamber kiln, shrinkage was also somewhat lower than in the hydrothermal kiln. Clay T2 shrinks, apart from in water vapour, stronger than Clay T1 and the test mixes during firing. The values are widely scattered and also virtually independent of the kiln atmosphere, in water vapour they are lower and in the chamber kiln higher than in the hydrothermal kiln. The clays differ in their composition. Clay T1 is a sandy clay and shows almost no firing shrinkage up to 890 °C. Clay T2 is a schistous clay, contains less quartz as stabilizing grain and has a higher flux content as well as coarse grain content < 250 µm. On account of its texturing, the measured values are widely scattered.
The bending strengths of the clays and mixes differ only insignificantly in the kiln atmospheres and different types of kiln with the exception of the water vapour atmosphere. For Clay T2 and the two mixes, this led to higher strengths. The values of Clay T2 were also scattered widely. The clay contain platey clay minerals like illite and muscovite, which lead to texturing and defects in the microstructure. In addition, on compaction and firing, they form a denser, stronger matrix. Higher strengths than in the hydrothermal kiln were reached by Clay T2 in the chamber kiln. Clay T1 on the other hand is loamy with a higher quartz content and fewer plastic clay minerals and leads to a homogeneous structure with similar strengths.
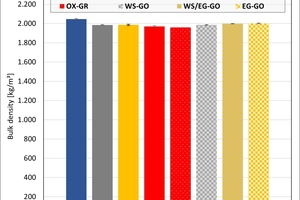
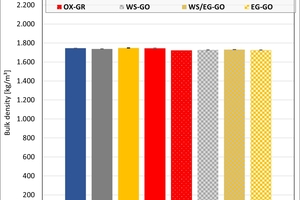
The bulk density of the clays ranged between 1 950 and 2 050 kg/m³. On account of their porosification, the mixes exhibited lower densities than the clays. The results show that the kiln atmosphere like the type of kiln had no significant influence on the density. Only in the firing with water vapour were slightly higher values determined in the clays.
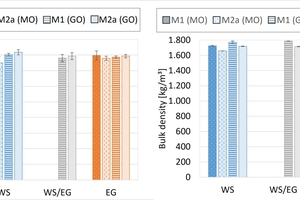
The results of the thermal conductivity and the bulk densities of Mixes M1 and M2a after firing in the hydrothermal and chamber kilns are shown in »8. The values are more scattered after firing in the hydrothermal kiln than after firing in the chamber kiln as these plates were ground with less precision.
The thermal conductivity of the mixes is slightly lower after firing in the hydrothermal kiln and slightly higher in the chamber kiln than for natural gas. The thermal conductivities in the chamber kiln are for 50 % hydrogen in the natural gas equal to the values for natural gas. The thermal conductivities are almost independent of the bulk density of the material, the values lie close to each other. In detail, however, it can be observed that with increasing bulk density, the thermal conductivity also increases.
The bulk densities of the mixes are almost independent of the kiln atmosphere. In the chamber kiln, the bulk densities were slightly higher than in the hydrothermal kiln. The bulk densities of Mix M1 are slightly lower than those of Mix M2a.
To summarize, it can be established that the kiln atmosphere formed with the combustion of hydrogen does not influence the ceramic properties of the tested materials both in the electrically and gas-heated chamber kiln in comparison with natural gas. This is shown for the simulated tunnel kiln atmosphere as well as the direct heating with hydrogen and natural gas. Although on account of the intermittent firing process in the gas-heated chamber kiln, much more water vapour is formed than in a tunnel kiln, no significant differences were observed in the fired properties. The results of the firing with 50 % hydrogen in the natural gas showed no differences to the firings with the individual gases. Only the water vapour atmosphere led to stronger sintering of all materials, which was shown in the higher mass losses, shrinkage, bulk densities, and strengths as well as lower water absorption values, in combination with a grey colouring.